 Assume that there are two heat reservoirs R 1 and R 2 at temperatures T 1 and T 2 (such as the stove and the block of ice). 
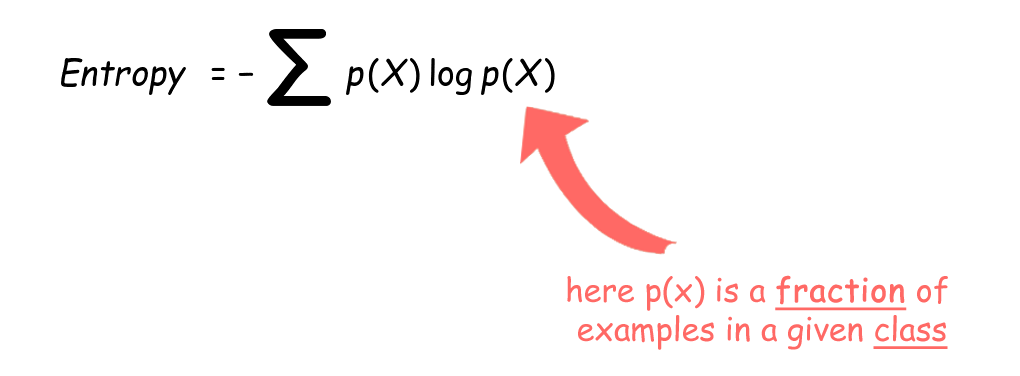
This equation effectively gives an alternate definition of temperature that agrees with the usual definition. For example, the block of ice and the stove constitute two parts of an isolated system for which total entropy increases as the ice melts.īy the Clausius definition, if an amount of heat Q flows into a large heat reservoir at temperature T above absolute zero, then the entropy increase is Δ S = Q/ T. The Clausius form of the second law states that spontaneous change for an irreversible process in an isolated system (that is, one that does not exchange heat or work with its surroundings) always proceeds in the direction of increasing entropy. To provide a quantitative measure for the direction of spontaneous change, Clausius introduced the concept of entropy as a precise way of expressing the second law of thermodynamics. For reversible processes the system is in equilibrium with its environment, while for irreversible processes it is not.Įntropy and the arrow of time See all videos for this article The latter process is reversible because only a slight increase in the restraining force could reverse the direction of the process from expansion to compression. Similarly, compressed gas confined in a cylinder could either expand freely into the atmosphere if a valve were opened (an irreversible process), or it could do useful work by pushing a moveable piston against the force needed to confine the gas. Such a process is reversible because only an infinitesimal amount of heat is needed to change its direction from progressive freezing to progressive thawing. In contrast, a block of ice placed in an ice-water bath will either thaw a little more or freeze a little more, depending on whether a small amount of heat is added to or subtracted from the system. Such a process is called irreversible because no slight change will cause the melted water to turn back into ice while the stove grows hotter. For example, a block of ice placed on a hot stove surely melts, while the stove grows cooler. The idea of entropy provides a mathematical way to encode the intuitive notion of which processes are impossible, even though they would not violate the fundamental law of conservation of energy. Its introduction by the German physicist Rudolf Clausius in 1850 is a highlight of 19th-century physics. The concept of entropy provides deep insight into the direction of spontaneous change for many everyday phenomena. Because work is obtained from ordered molecular motion, the amount of entropy is also a measure of the molecular disorder, or randomness, of a system.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
